Blog article - 26.01.2021
evonos can be your perfect fit as a medical device company. Here are six reasons why.

Around the world, there are hundreds of medical device companies providing hospitals and clinics with the equipment they need. But not all medical device companies are the same; they vary by approach, by quality, by ethos. Given the critical nature of the work done by hospitals, and the care given to every patient, purchasing departments and surgeons give great thought to the suppliers they choose.
What are the qualities you need to look for in a medical device company?
What company qualities make a difference to the medical equipment they sell, which in turn make a difference to the level of care you provide? Here, we look at six factors that separate medical device companies – and what you should look for in yours.
Who are the top medical device companies?
Everyone knows the big names in the medical device industry. By virtue of their size it is easy to see them as the providers of the highest quality medical equipment – but choosing a supplier is not that simple. You should be asking what companies make medical equipment that suits your needs. A smaller, specialist provider can deliver better designs or a more cost-effective option. Sometimes, however, it is not about company size at all.
Whether your chosen supplier is a multi-national or a niche local player, these are the qualities to look for – and the questions to ask.
1. Do medical companies actually talk to surgeons?
Most hospitals and healthcare providers do their buying through a purchasing department – but the end-customer is the surgeon or clinical care giver. If a global medical device company is going to succeed, it must therefore work to understand their needs. And this will not happen unless the company gets qualified industry feedback and input from its ultimate target audience.
Medical device companies can achieve this in a number of ways – if your industry supplier engages in any of the following activities, it’s a good sign that their devices will be effectively designed and bring you results:
- conducting regular survey research into surgeons’ needs
- collaborating with surgeons – involvement in NPD (new product development) sessions
- gathering formalized feedback on company and product experience
- nurturing personal relationship with surgeons, through discussions and invitations to industry events etc
We also invite surgeons to take part in discussions at the early stages of new product development. This “blue-sky-thinking” stage is hugely important and allows surgeons to contribute thoughts and ideas of any kind – nothing is too ambitious – to our design team. Armed with this input, we can then investigate whether a certain feature can be cost-effectively incorporated into the design of the next medical devices.
One of the reasons evonos has been so successful is that we try to listen carefully to feedback from surgeons, and this is the starting point for a number of medical device design innovations that lead to higher-performing surgeons and better patient care. The design of the evoDrill perforator, for example, was guided by the realization that surgeons sometimes need to use significant physical force to penetrate the cranium.
Every neurosurgeon is capable of exerting that force, of course, but we understood that, with an increasing number of women working in the field, we needed to design a cutting edge that required less brute force and allowed more precision. Even the smallest amount of fatigue can impact on performance. The evoDrill cuts in a way that simply requires less downward force; its design is a reflection on the needs of today’s surgeons, and is a key product feature that has made our perforator so popular in the industry.
2. How well do medical device manufacturers understand hospital processes?
It is one thing understanding the surgeons – but it is also important for a medical instrument manufacturer to understand the processes used both inside and outside the operating theatre.
One important area concerns the way that devices are sterilized. Re-usable perforators, for example, can be used for around 10-15 operations. For the first three, the performance is excellent, but it gradually degrades over time as the drill loses its edge. Single-use perforators are always perfectly sharp, but there may appear to be a trade-off in terms of cost.
However, once you start to look at the expensive processes involved in cleaning and sterilizing (and re-sharpening) equipment, you realise there is a cost there too. It also takes time, which often means holding a number of re-usable perforators in the inventory, which adds further to the capital investment.
It is this understanding that has led evonos to prefer single-use perforators. In our opinion, and it is a view shared by customers, single-use medical devices with a guarantee of sharpness compare very well in terms of the total cost of ownership.
3. Does your chosen medical instrument company invest in the newest technology?
The design of medical devices is constantly evolving. As new designs appear, they gradually replace inferior, older designs in order to progressively improve both the surgeon experience and the patient outcome. However, investment in the latest manufacturing technology is expensive and it is understandable that some medical device manufacturers prefer to mass-produce less innovative products. There are weaknesses in the design of these products, but a below-average price may compensate for the below-average performance.
The technology used by evonos for our patient specific evoShape cranial implants is a case in point. We use 5-axis milling technology based on high-resolution CT medical imaging data in order to create an implant that fits perfectly. Other techniques such as casting or pressure welding techniques are used to produce implants and these are used all over the world.
However, the accuracy provided by our milling technology simply creates a better fit – which in turn makes the process easier for the surgeon and causes less postoperative irritation for the patient, along with a more pleasing aesthetic result. We are already looking into the next evolution of technology in the form of 3D-printing in order to improve the quality of our products even further.
4. Is your chosen medical supplier driven by the desire to innovate – or by shareholder profit?
But why do some medical companies look to innovate, while industry players prefer to scale down the investment in new technology and back the commercially safe option? One important factor is the structure of the company itself. A smaller, newer medical company may be owned and operated by a small number of people who are personally involved in customer dialog and product innovation. With larger companies, this is not possible. As the hierarchy increases there is a bigger gap between customer and product design.
There is also a difference in terms of ownership. A larger, publicly listed company has shareholders to satisfy. Long-term investment in innovation is more difficult to justify when the industry insists on regular dividends. The largest medical company will have greater economies of scale of course – but does it really outweigh the importance of that freedom to bring new device designs to market, which is essential to drive better device performance?
5. The best medical device company is the one with the best people
A medical company is only as good as its people; the best devices are designed by the best minds, supported by the best managers in the industry. But how can you judge the quality of the people employed by your chosen supplier?
Almost all of the highest-quality and most innovative medical device companies are started by people with personal industry experience. The founders are often former surgeons, clinical staff or device design experts who have decided to do things differently. This combination of entrepreneurialism, supported by first-hand knowledge of how the products are used makes a huge difference.
These people understand the needs of their target audience because they have done their job themselves. The medical product weaknesses that held them back, the difficulties they faced, the problems that affected patient outcomes – these are industry experiences that drive them to do better – and are clear to anyone dealing with those companies.
At evonos, we think it is very important to ensure that our employees feel comfortable and have the chance to improve themselves further. We try to involve our employees across all departments in everyday activities and offer rewarding working conditions. Because we know: happy employees are motivated employees.
6. Do surgical device companies from different countries have different approaches?
Bear in mind also that medical manufacturing practices and product designs vary across the world, and it is typical to see different approaches applied in different countries. Chinese manufacturers, for example, are highly skilled in lean manufacturing processes. Add this to the economies of scale achieved in some of the largest industry facilities in the world that are found in China, and you have a highly efficient operation – which can drive down the unit cost of each product. Chinese devices can be very high quality too, but they are renowned for their manufacturing efficiency and therefore affordability.
By contrast, US manufacturers are the marketing experts. They are very good at communicating product benefits and building relationships with hospitals and healthcare providers. European manufacturers, especially German manufacturers have built a reputation for the very highest standards of engineering. With higher labour costs, they are unlikely to bring to market the cheapest option, but standards of quality and manufacture are often extremely high. At evonos you can 100% rely on quality made in Germany.


 evoDrill
evoDrill evoFix
evoFix evoFix Sterile
evoFix Sterile evoBase
evoBase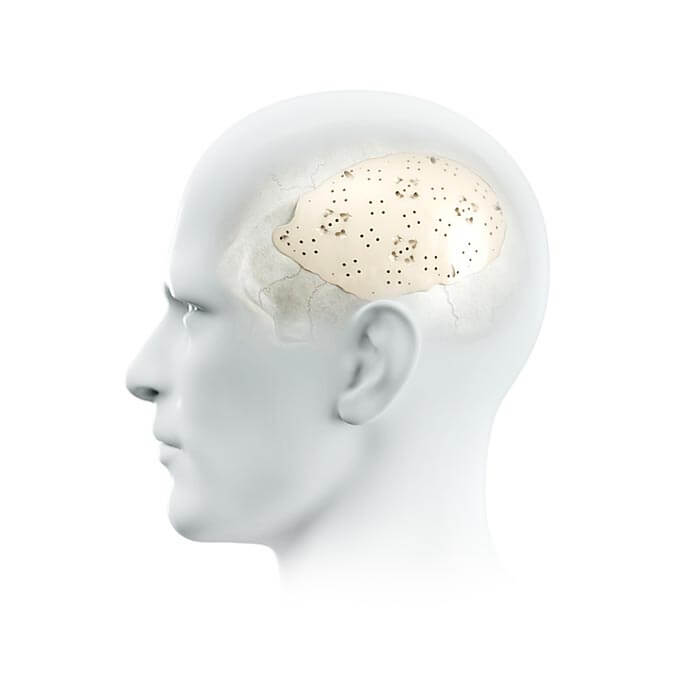 evoShape
evoShape evoLight
evoLight evoClip
evoClip evoFrame
evoFrame evoBite
evoBite evoCarat
evoCarat